
Investment thesis
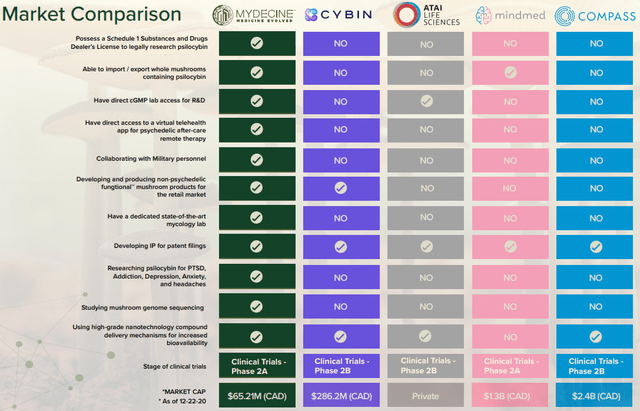
I like the psychedelics sector because the regulatory tide seems to be turning. However, there seems to be little coverage of the companies from this field on SA and I think the reason for this is that many of them are based in Canada. Well, I’ve already covered Numinus Wellness (LKYSF) and Cybin (CLXPF) and today I’m taking a look at Mydecine Innovations Group (OTCPK:MYCOF). The latter has several advantages over its competitors such as collaborating with military personnel as well as access to a mycology lab. The company also has a license to legally research psilocybin and it owns a virtual telehealth app for psychedelic after-care remote therapy.
Just like Numinus and Cybin, Mydecine is another narrative-driven stock in the psychedelics space and I like the story behind it.
Overview of the business
In its own words, Mydecine is an emerging biotech and life sciences company dedicated to developing and commercializing innovative solutions for treating mental health problems and enhancing vitality.
What does this mean? Well, in layman’s terms, the company plans to use magic mushrooms to treat posttraumatic stress disorder (PTSD). Yes, psilocybin is found in magic mushrooms. Mydecine has seven molecule designs and its clinical trials are at Phase 2A, with test sites approved at Leiden University, University Of Alberta, Royal Ottawa Hospital, and Western Ontario University. Overall, it’s at a pretty early stage but it has developed partnerships and secured licenses that provide it with several advantages over its peers.
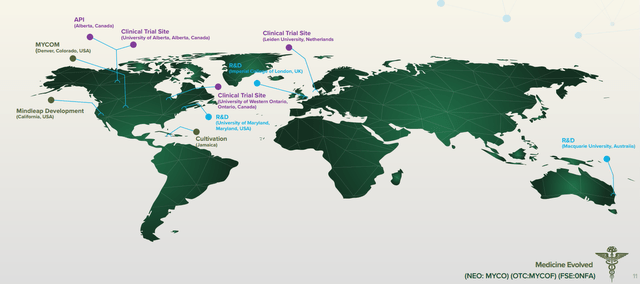
(Source: Mydecine)
It’s too early to tell if any of the molecules can lead a commercial success.
In December 2020, Mydecine completed a commercial harvest of 20 kilograms of magic mushrooms and in March 2021 it became the first company in the world to complete an international legal export of dried magic mushrooms.
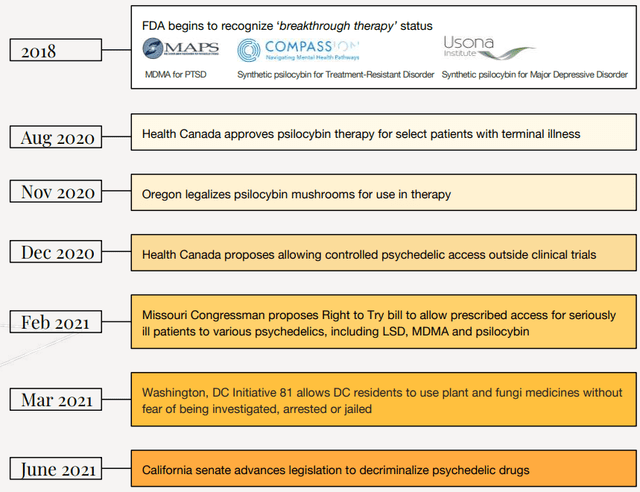
Notice how I keep mentioning legal. This is because psychedelic-assisted therapeutic solutions companies are in limbo at the moment. Some of the leaders in the sector like COMPASS Pathways (CMPS), MindMed (MNMD), and Numinus have high valuations based on the eventuality that governments around the world will approve the therapeutic use of psychedelics. And as I mentioned at the start of this article, the tide is turning, especially in the USA.
In 2018, COMPASS’ synthetic psilocybin (COMP360) was granted breakthrough therapy status by the U.S. Food and Drug Administration. In June 2021, the Drug Policy Reform Act was introduced in Congress. It has the potential to become the first bill to federally decriminalize possession of all currently illicit drugs and this would likely lead to a loosening of restrictions on research into psychedelic therapies.
(Source: Numinus)
The mental health market opportunity for psychedelic-assisted therapeutic solutions is estimated to be pretty large. According to data from the National Institute of Mental Health, the…
Read more:Mydecine’s Prospects Look Magical, Like A Mushroom (OTCMKTS:MYCOF)









 Bitcoin
Bitcoin  Ethereum
Ethereum  Tether
Tether  Solana
Solana  USDC
USDC  Lido Staked Ether
Lido Staked Ether  XRP
XRP  Dogecoin
Dogecoin  Toncoin
Toncoin  Cardano
Cardano  Shiba Inu
Shiba Inu  Avalanche
Avalanche  TRON
TRON  Wrapped Bitcoin
Wrapped Bitcoin  Bitcoin Cash
Bitcoin Cash  Polkadot
Polkadot  Chainlink
Chainlink  NEAR Protocol
NEAR Protocol  Polygon
Polygon  Litecoin
Litecoin  Internet Computer
Internet Computer  Uniswap
Uniswap  LEO Token
LEO Token  Dai
Dai  First Digital USD
First Digital USD  Hedera
Hedera  Ethereum Classic
Ethereum Classic  Stacks
Stacks  Aptos
Aptos  Mantle
Mantle  Cronos
Cronos  Stellar
Stellar  Render
Render  Cosmos Hub
Cosmos Hub  Filecoin
Filecoin  OKB
OKB  Pepe
Pepe  Renzo Restaked ETH
Renzo Restaked ETH  dogwifhat
dogwifhat  Immutable
Immutable  Bittensor
Bittensor  XT.com
XT.com  Arbitrum
Arbitrum  Maker
Maker  Wrapped eETH
Wrapped eETH  Optimism
Optimism  The Graph
The Graph